Monochromator.us
Let there be light! Fiat lux!
Color wars
Here in the northeastern USA many of us put feeders for the birds, especially in the winter. Watching the birds at the feeder is engaging and loaded with physics and spectroscopy too.

According to what I read the WSJ last weekend. In birds there are (at least) two types of color, pigmented and structural. The cardinal is red because its feathers really are red. They are loaded with pigments called carotenoids. These pigments are also responsible for the gold in goldfinches and the yellow in yellow warblers. They are the reason carrots are orange.
Blue and green are oddball colors in the animal kingdom. They are structural colors. In birds, they are caused by microscopic structures in the feather that refract light like a prism. You can test this yourself. The next time you find a blue jay feather, hold it up against a bulb so that the light passes through it. Or crush the feather, destroying the intricate structures. In both cases, the blue disappears.
Read more about this backyard physics at “What color is a blue jay? and ““The Color War of Blue Jays and Cardinals”
Let there be light
Clean, safe and unlimited power has been an obsession for scientists and inventors for centuries, and an underlying preoccupation for our society as a whole. Since the 1940s, when we first understood how the sun creates energy, nuclear fusion has been energy’s holy grail. Check out © 2017 EyeSteelFilm

XUV spectrometers with 64-bit LightField software
McPherson and Princeton Instruments are pleased to announce introduction of a new Add-In for Princeton Instruments’ powerful 64-bit LightField software that seamlessly controls McPherson’s deep UV and soft X-ray spectrometers and Princeton Instruments soft x-ray cameras. This Add-In allows users to keep all the benefits of LightField including easy setup and access to exclusive features like the real-time math engine, SmartSearch, and the virtual optical bench for conveniently managing multiple experimental setups.
- 1-100nm Spectrograph w PIXIS-XO
- 5-300nm Monochromator+Spectrometer w PIXIS-XO
- 50-550nm Monochromator+Spectrometer w PIXIS-XO
- Copy of the press release
Contact McPherson to learn how Princeton Instruments detectors integrate for your application and how LightField controls the hardware. McPherson vacuum compatible instruments work from less than 1 nanometer with a selection of diffraction gratings. McPherson’s flat field, conical dispersion and other specialized solutions are available today, please inquire.
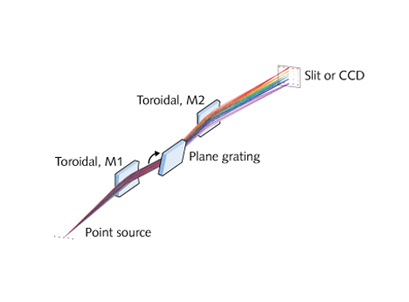
Off-plane and conical dispersion spectrometer
For operation from approximately 8 to 125 nm (10 to 150 eV), the OP-XCT can operate as a monochromator or imaging spectrograph. Illumination from a point source strikes a toroidal mirror (to reduce optical aberrations) at 85° angle of incidence. This grazing angle improves reflection efficiency at short wavelengths. Light passes to a triple turret plane grating (three user-specified blaze wavelengths/groove densities can be selected) inside a vacuum vessel and then to a toroidal mirror before passing the exit slit.
Off-plane instruments have significantly higher diffraction-grating efficiency than on-plane designs; experimental results have demonstrated high efficiencies follow modeled results. The plane gratings used in the OP-XCT are available with many different blaze angles and spectral resolution is a function of the gratings selected by the user and the entrance slit or source size. Focal length also directly influences attainable resolving power. With a 0.05 mm source and a CCD detector, the 800-mm-focal-length OP-XCT resolving power (λ/Δλ) measures higher than 300 at 25.6 nm (48 eV).
Placing a 2D detector at the exit allows the off-plane spectrometer to work as an imaging spectrograph. With a coarse diffraction grating, it can survey a broad wavelength swath from 10 to 140 nm on a detector only 12 millimeters wide.
Learn more about this instrument in the article "2D Imaging Spectrometer for EUV and Soft X-ray" from McPherson and the one that appeared in Laser Focus World
Water window
Spectrometers currently are available for much of the soft x-ray, extreme UV and VUV regions, although not always in the form in which we are accustomed to seeing. Selecting a spectrometer requires prioritizing analytical goals, wavelength range, spectral resolution, dispersion and possibly optical aperture requirements in light of experimental needs. Shorter wavelengths require more care in selection of optical schemes to maintain efficiency and performance. We wrote up a brief description about this for the Photonics Spectra magazine in 2011.
As example, the grazing incidence Rowland circle spectrometer Model 248/310 made some of the measurements described in this technical paper "Water-window soft x-ray high-harmonic generation up to the nitrogen K-edge driven by a kHz, 2.1 μm OPCPA source.” As far as I know, the experimental work done in this paper comes from Department of Electrical Engineering and Computer Science and Research Laboratory of Electronics, Massachusetts Institute of Technology. There is strong collaboration with Grupo de Fotônica, Universidade de São Paulo, Brazil as well as Center for Free-Electron Laser Science, DESY, the Department of Physics, University of Hamburg, and the Hamburg Center for Ultrafast Imaging, Germany
Spectroscopy as a Scientific Tool
The accomplishments achieved by scientists through use of the spectroscope form a list so imposing as to leave no doubt that this instrument is one of the most powerful now available for investigating the natural universe. But spectroscopy is valuable not only to the research scientist; it finds everyday and increasing use in technological laboratories. Today directors of such varied enterprises as factories, assay offices, arsenals, mines, crime detection bureaus, public health departments, hospitals, museums, and technical research institutes consider access to spectroscopic equipment essential to proper functioning of their laboratories.
THE SPECTRUM | A spectrum has been defined as the ordered arrangement of radiation according to wavelength. Electromagnetic radiations have been discovered that have wavelengths of every value in the range from thousands of kilometers to trillionths of a millimeter. A complete electromagnetic spectrum would comprise all these radiations arranged to order from the longest to the shortest wavelengths. Since no single instrument exists that will separate all radiation containing all these wavelengths into a spectrum, the electromagnetic spectrum has been divided into “regions” in accordance with the types of instruments available to produce and detect the waves of various lengths.
From? idk. Attribution unknown. Probably a spectroscopy book published during first half of the twentieth century. These are opening paragraphs of Chapter One
Light
LIGHT. This term properly refers to the range of electromagnetic radiation frequencies associated with vision; though the physicist is apt to think of more objective manifestations, such as the photovoltaic effects, and sometimes even oversteps the limits of the visible by calling infra-red or ultra-violet radiation, and even x-rays “light”. The wavelengths of visible light extend approximately from 400 nanometers (extreme violet) to 770 nanometers (extreme red). Compared with radiation as a whole this is an extremely limited range. It appears to be an inevitable limitation, however, on account that of the strong absorption of most substances for radiation on both sides of it. The quantum theory of radiation applies of course to light, the energy quanta of which are called photons. Some of the optical phenomena so readily interpreted on the wave theory, such as reflection, refraction, interference, and polarization of light offer difficulties when studied in terms of quanta; the laws of photoelectric phenomena, photoconductivity, the spectrum, etc. on the other hand become much more intelligible. Geometrical optics is easily expressed in terms of either. The well-known Huygens principle and Fermat principle apply to any radiation, including light, as does the electromagnetic constant representing the speed of light in a vacuum. Discussions of photometry and of color are, however, usually in terms of visual sensation and hence are confined to light proper, though the physical ideas involved are bit subject to limitation.
From the entry for “light” in Van Nostrand’s Scientific Encyclopedia
Thorium nuClock
The work with Thorium seems really provocative; it may help to realize new vacuum ultraviolet spectra from transitions and also to time keeping on whole new level. One group, nuClock.com has been working in this direction and has a new publication. "We propose a simple approach to measure the energy of the few-eV isomeric state in 229Th. To this end, 233U nuclei are doped into VUV-transparent crystals, where they undergo α decay into 229Th, and, with a probability of 2%, populate the isomeric state. These 229mTh nuclei may decay into the nuclear ground state under emission of the sought-after VUV γ ray, whose wavelength can be determined with a spectrometer." Read the whole abstract and more at Phys. Rev. C journal.
---------------------------
McPherson has standard vacuum ultraviolet spectrographs for detection of transmission, relfection and emission in the 100 to 500 nanometer wavelength range. The 234/302 is a good example.
---------------------------
Thermonuclear fusion science
This just in.... "We are pleased to inform you that National Institute for Fusion Science is going to organize the joint meeting of 26th International Toki Conference and 11th Asia Plasma and Fusion Association Conference. The international program committee chaired by Dr. Yutaka Kamada of National Institute for Quantum and Radiological Science and Technology and the local organizing committee have started to organize the conference.
For further information, the conference web page

Contribute to the conference and the progress of this field!
See also Plasma and Fusion Research an electronic journal published by The Japan Society of Plasma Science and Nuclear Fusion Research. The journal, covering the wide disciplines of plasma science and technology, is intended to provide an accessible and comprehensive source for analysis and contemporary information and data in all areas of plasma and fusion research.
---------------------------
McPherson has extreme ultraviolet spectrographs that can collect a wide wavelength range in one acquisition. There are different gratings for soft x-ray (SXR, XUV), extreme ultraviolet (EUV), and VUV. The 251 SPRED is a practical instrument for applications requiring rapid data collection from transient, short lived events like fusion plasmas in tokamak devices, laser ablation, liquid droplet EUVL sources, and more.
---------------------------
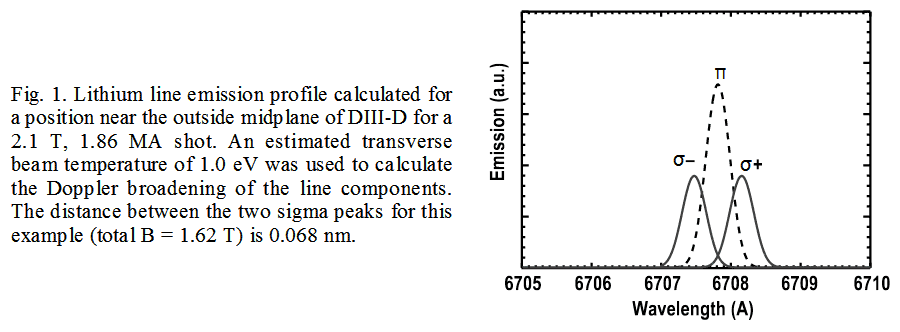
High resolution techniques and visible light diagnostic doppler broadening, Stokes and Zeeman effects work with the McPherson 209 spectrometer. Well equipped it can simultaneously monitor spectra from 100 input fibers on one commercial 1024*1024 pixel, ~13mm square CCD. Sub Angstrom spectral resolution is attained in all channels and ideally suited for simultaneously intercepting and analyzing multiple spectra from spatially distinct regions.
---------------------------
Iridium for VUV
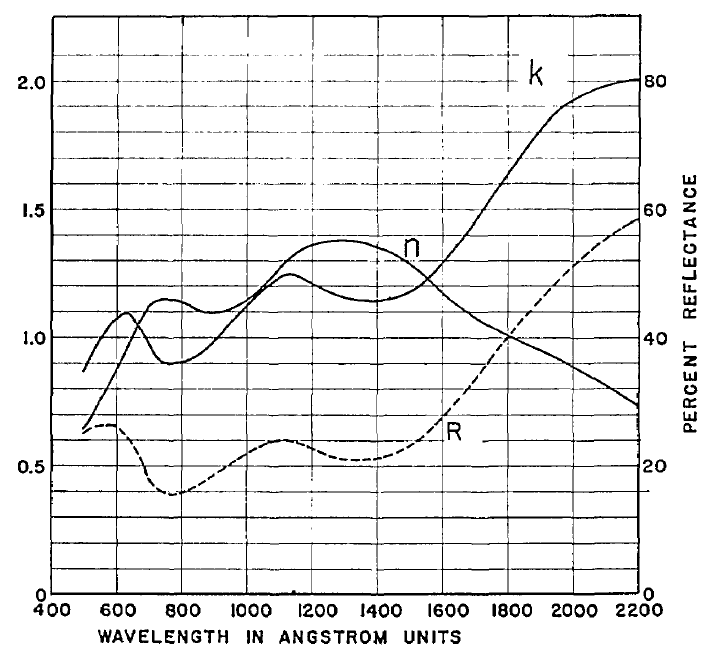
The reflectance and optical constants of evaporated iridium films were measured* in the wavelength region from 500 to 2000 Å. The films were evaporated by electron bombardment and deposited at various rates onto glass substrates of various temperatures. Their optical constants were determined from reflectance measurements made at different angles of incidence. In contrast to aluminum and other oxide-film-forming metals, iridium showed very little change in reflectance during exposure to air. Films with highest normal incidence reflectance were obtained by evaporation onto substrates heated to 300°C and more. Owing to interference, semitransparent films of 100 to 200 Å on glass showed higher reflectance than opaque films at most wavelengths in the vacuum ultraviolet. Iridium films produced under optimum conditions had a reflectance of about 27% at 55 nanometers
*The Ir mirrors were transferred in air to a reflectometer where their reflectances could be measured at any angle of incidence between 60 and 86-degrees. A photomultiplier sensitized with sodium salicylate was used as the detector. A normal incidence vacuum-uv grating monochromator was employed to cover the spectral range from 500 A to 2200 A, and a grazing incidence grating monochromator was used to obtain reflectance data at 304 A. Two types of light sources were used to make measurements over the entire wavelength region. One was a dc glow discharge and the other a pulsed discharge source. [similar to hollow cathode]
More at: Optical Properties of Evaporated Iridium in the Vacuum Ultraviolet from 500 Ångstroms to 2000 Ångstroms by G. Hass, G. F. Jacobus, and W. R. Hunter. JOSA 57, 6, 758. 1967
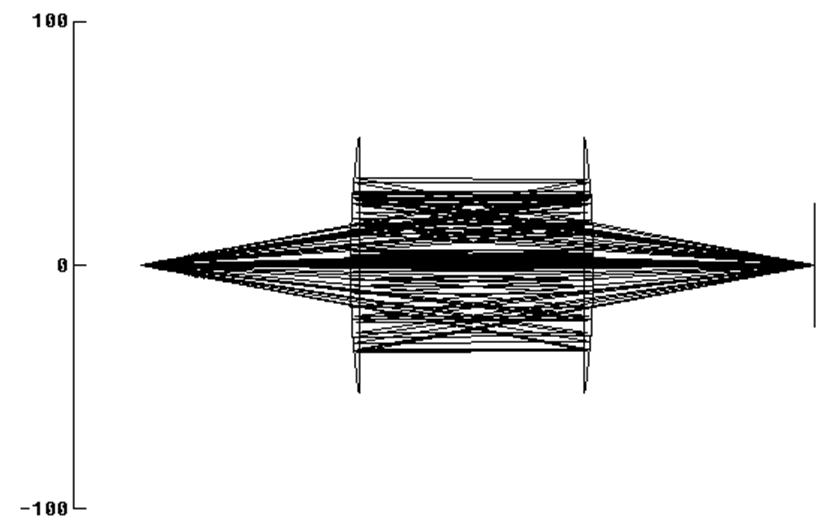
Yolo objective
Tonight I’m reflecting on Chapter 18 of the Handbook of Optics tonight “Reflective and Catadioptric Objectives” (Lloyd Jones, Optical Sciences Center, University of Arizona, Tucson). Commercial two mirror reflective objectives work at infinity or magnifications ranging from 15 to 40X. Companies like Companies like Newport, Beck/Ealing, Thorlabs and Edmund sell these. These designs generally use two spherical mirrors to collect an aggressive solid angle. Mirrors with high performance deep ultraviolet Al+MgF2 work at Lyman-Alpha and generally above 110 nanometers. The obscuration and central hole reduces contrast in the final image. Because of the different sizes of the optics magnification will be at least about 4X. The high magnification offered commercially helps performance specifications. The designs are compact that can help or hinder deployment in through vacuum flange applications. Basically these are classic Schwarzschild designs corrected for third-order spherical aberration, coma, and astigmatism, resulting in near-diffraction-limited performance.
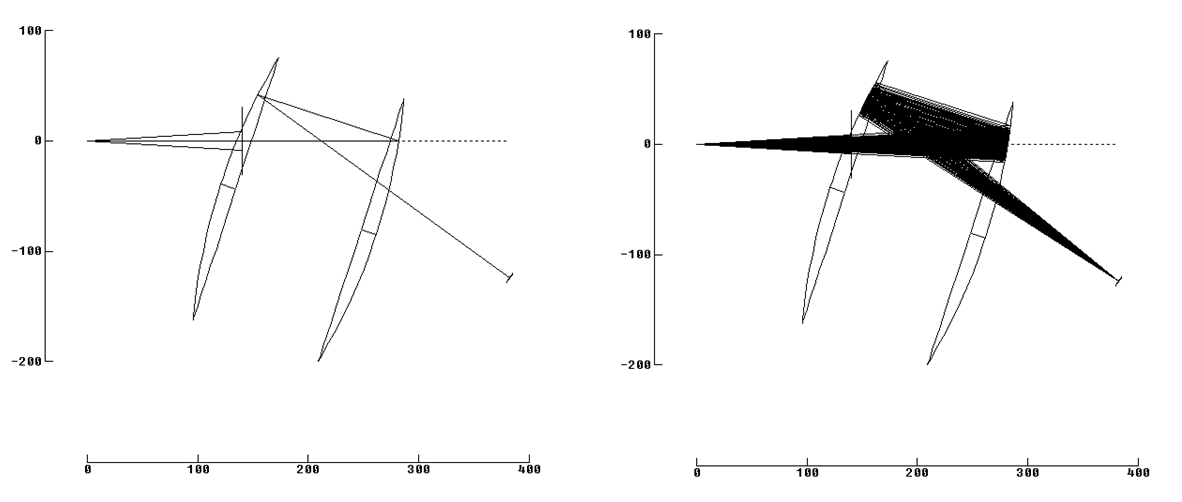
We build a low magnification reflective objective similar to Schwarzschild. A reflective objective for Prof. Dr. Michael A. Rübhausen (Uni Hamburg, Germany) uses parabolic optics arranged to face one another. This design has central hole (obscuration) and if used as camera objective, the camera looks directly at the source.
Parabolic optics provide low 1:1 magnification. Imaging is superior to the Schwarzschild over a small field. Still the obscuration (central hole) reduces contrast in the final images. Carefully introduced baffles can prevent line of sight from detector to source. For nomenclature, while mirror size is appreciably different, the arrangement of optics and selection of conics is same as Gregorian telescopes.
Taking the low magnification and superior imaging of the Gregorian/Ruebhausen variant, and using an eccentric section, we can arrange two off axis parabolic optics in a “Yolo” configuration (by A.S. Leonard and named after a scenic county in California). The resulting design is good for work at slower aperture ratios so can “look” through vacuum port holes more effectively, has no obscuration for improved contrast, has excellent imaging over small fields and uses relatively common and accessible aspheric optics (off axis parabolas.) Cost is high but mirrors like this are routinely built and testable.
Preamble done, we arrive at a vacuum ultraviolet capable eccentric Gregorian / Yolo objective design. No central hole or obscuration improves contrast. Two reflective surfaces maintain high throughput (reflective efficiency) even in the vacuum and deep ultraviolet. Commercially available optics used for improved cost and delivery time. About 5 micron images from point source over 2mm field). Source to focal plane distance is about 400mm overall, for better fit with vacuum chambers and cameras. Camera does not have line of sight towards source.
Reach out to us if you need something like this for your experiments.
Lots more in Chapter 18 of the Handbook of Optics tonight “Reflective and Catadioptric Objectives” (Lloyd Jones, Optical Sciences Center, University of Arizona, Tucson).
Compact extreme ultraviolet source
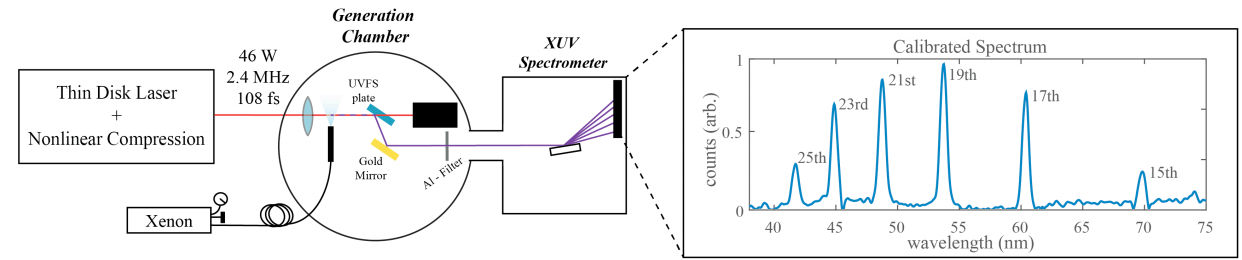
A research group at ETH Zürich's (Dr. Florian Emaury) Institute of Quantum Electronics demonstrated, for the first time, HHG driven by the compressed output of a thin-disk oscillator. The demonstration opens the door to more compact oscillator-driven Extreme-UV, soft x-ray and XUV radiation at high rep rates. Further ongoing steps to increase the flux of the novel source include better pressure handling and pulse duration reduction. The demonstrated source provides a starting point for a broad range of applications in which high repetition-rate low-noise XUV pulses are crucial. These include ultrafast photoemission studies on solid surfaces or coincidence detection schemes in atomic, molecular and optical physics
High-harmonics generation experiment using compressed output of a thin disk laser. In the generation chamber, the compressed pulses are focused on a 22 µm spot diameter to reach an intensity of ≈ 5.5x10E13 W/cm2. The XUV light generated is distinguished from the infrared light via reflection on a fused silicate plate Brewster’s angle and then routed to the spectrometer by a focusing gold mirror. Our XUV spectrometer (McPherson 248/310G) enables to measure the spectrum of the XUV transmitted through a 400 nm Aluminum filter, showing generation down to the 25th harmonic (41.2 nm – 30.1 eV).
Want more? Follow the link to this research article in Optica "Compact extreme ultraviolet source at megahertz pulse repetition rate with a low-noise ultrafast thin-disk laser oscillator" by F. Emaury, A. Diebold, C.J. Saraceno, and U. Keller and "Oscillator-Driven High Harmonic Generation" by F. Emaury, A. Diebold, C.J. Saraceno, and U. Keller
Better LEDs From Salmon Sperm
A University of Cincinnati researcher has an unusual approach to developing “green” electronics — salmon sperm. Professor Andrew Steckl, a leading expert in light-emitting diodes, is intensifying the properties of LEDs by introducing biological materials, specifically salmon DNA.
Read the whole paper here
Electrons move constantly — think of tiny particles with a negative charge and attention deficit disorder. It is through the movement of these electrons that electric current flows and light is created. He believed that if the electrons’ mobility could be manipulated, then new properties could be revealed...“Biological materials have many technologically important qualities — electronic, optical, structural, magnetic,” says Steckl. He also wanted a source that was widely available, would not have to be mined, and was not subject to any organization or country’s monopoly. His answer? Salmon sperm.
More here about Better LEDs From Salmon Sperm
Magnetism and Light
Following are some excerpts I enjoyed from
Magnets: The Education of a Physicist (Book by Francis Bitter, 1959)
https://www.amazon.com/Magnets-Education-Physicist-Science
Magnets: The Education of a Physicist
Atoms and Molecules
MAGNETISM AND LIGHT
“I made a series of measurements and found that one of the subtler predictions of quantum theory regarding peculiarities in the susceptibility of NO was verified experimentally … a student of the sun, to attempt to measure the magnetic field on the sun by spectroscopically analyzing light emitted from its various parts … a chance to study a new aspect of magnetism involving a very peculiar aspect of quantum theoretical predictions”
“I began to study the quantum theory of light and the Zeeman effect, as the effect of a magnetic field on the emission of light from an atom is called. An atom that radiates certain particular colors or frequencies in the absence of a magnetic field radiates a more complicated spectrum when a magnetic field is turned on. Each of the individual spectral lines, or each of the individual frequencies which an atom radiates, splits up into two or three or more. The splitting is usually only very slight and requires most sensitive apparatus to detect it, but it is there, and it takes quantum theory to explain it.”
“The quantum theory said … an electron could move around in an atom at certain frequencies without radiating at all…The state with the least energy—the one in which the electron moves around the fastest—is the stable state. In this state it cannot radiate at all.”
“But if atoms are violently bombarded either by collisions in a gas at very high temperature as in the sun, or put in a vacuum tube in which electrons, accelerated by electric fields, make energetic collisions with atoms, they may be excited to one of the higher states. In these states of higher energy, or excited states, the atoms do not radiate either, but in making transitions to lower states, or states of lower energy, the get rid of some excess energy and this energy is radiated…the particular color or frequency of the light an atom radiates depends only on the total amount of energy which it gets rid of in a quantum jump from excited state to a lower state having less energy. The greater the jump, the bluer the light or the higher frequency; the smaller the jump, the more the color of the light radiated goes towards the red or lower frequency in the spectrum”
Read the whole paper here
“For the simplest atom of all, the hydrogen atom, in which a single electron moves around its nucleus, the exact elevation of [excited] steps can be calculated. It is possible to predict exactly from known quantities—namely the mass of the electron, the amount of charge on it, and the allowed values of the angular momentum—just what the height of the hypothetical steps are, and consequently just what colors a hydrogen atom should radiate.”
“A compass needle tends to set itself parallel to a magnetic field. If we attempt to turn it, we must push on it and increase its energy, the amount of increase depends on how far we turn it. But, since the orientations of the atomic magnets according to quantum theory are limited (for example, the hydrogen atom in its lowest or ground state can have only two), we must expect the magnetic field to split up the ground state into two, corresponding to the to the energy of the atom when its magnetism is parallel or anti-parallel to the field. Since the energy levels are split by a magnetic field, the frequencies radiated by transmission from and to these levels will be multiplied in number—hence the Zeeman effect referred to above.”
“…I was several hundred dollars in debt.’”
Excerpts from
Magnets: The Education of a Physicist (Book by Francis Bitter, 1959)
https://www.amazon.com/Magnets-Education-Physicist-Science
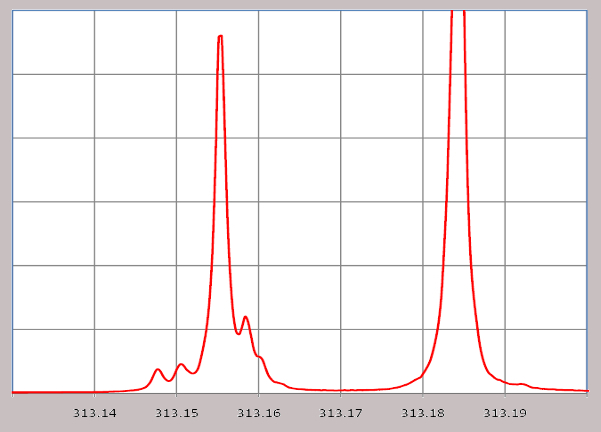
A high resolution spectrometer like the McPherson Model 209 is a most sensitive apparatus capable of detecting the slight wavelength shifts associated with Zeeman, Stokes and Doppler effects.
Happy New Year!
Some 2017 spectroscopy related events:
- 9th International Workshop on Microplasmas, June 6-9, 2017 - Garmisch Partenkirchen (Germany)
- 2016 EUVL Source Workshop Proceedings and First Call for Papers 2017 EUVL Workshop
- 44th Annual International Conference on Plasma Science (ICOPS 2017) will be May 21-25 in Atlantic City, NJ